|
Phosphates (PO 4 3-) are used in the production of special glasses, sodium lamps, in Production of steel, and some other applications, such as pyrotechnics, toothpaste, detergents (phased out in some countries), pesticides etc.īone ash (calcium phosphate, Ca 5(OH)(PO 4) 3) is used to create Fine chinaware and to produce mono-calcium phosphate (CaH 4P 2O 8), which is used in Baking powder. The largest use of phosphorus compounds (Concentrated phosphoric acids, which may contains 70% – 75% P 2O 5) are used in fertilizers for agriculture and farm production.Īmmonium phosphate ((NH 4) 3PO 4) is made from phosphate ores, where the ores are firstly converted into phosphoric acids (H 3PO 4) before being made into ammonium phosphate. Red phosphorus is used in the material (binder, powdered glass, & red phosphorus), which stuck on the sides of matchboxes, and when heat is generated by friction the red phosphorus convert to white phosphorus, which ignites spontaneously. White phosphorus is used in flares & military applications (incendiary bombs, smoke bombs, tracer bullets, smoke screening etc.). Recognised as an element: Antoine Lavoisier (1777) Phosphorus Uses Naming: Greek: phôs (light) & phoros (bearer)ĭiscovery: Hennig brand (1669), Hamburg (Germany) 
In solube in water as well as in Organic compounds Insoluble in water but soluble in Organic compounds Obtained by heating White phosphorus (250 0 C) When white is ignited with charcoal, calcium metaphosphate yields 2/3 rd of white phosphorus and 1/3 rd is remain in the residue as calcium orthophosphate:ģ Ca(PO 3) 2 + 10 C → Ca 3(PO 4) 2 + 10 CO + P White Phosphorus VS Red Phosphorus In this reaction Monocalcium phosphate is dehydrated to the corresponding metaphosphate: In this reaction, Tricalcium phosphate (Ca 3(PO 4) 2) in bone ash is converted to monocalcium phosphate (Ca(H 2PO 4) 2) with sulfuric acid (H 2SO 4):Ĭa 3(PO 4) 2 + 2 H 2SO 4 → Ca(H 2PO 4) 2 + 2 CaSO 4 
Reaction of Another process by which phosphorus is extracted by applying high temperature (1500 o C):Ģ Ca 3(PO 4) 2 + 6 SiO 2 + 10 C → 6 CaSiO 3 + 10 CO + P 4 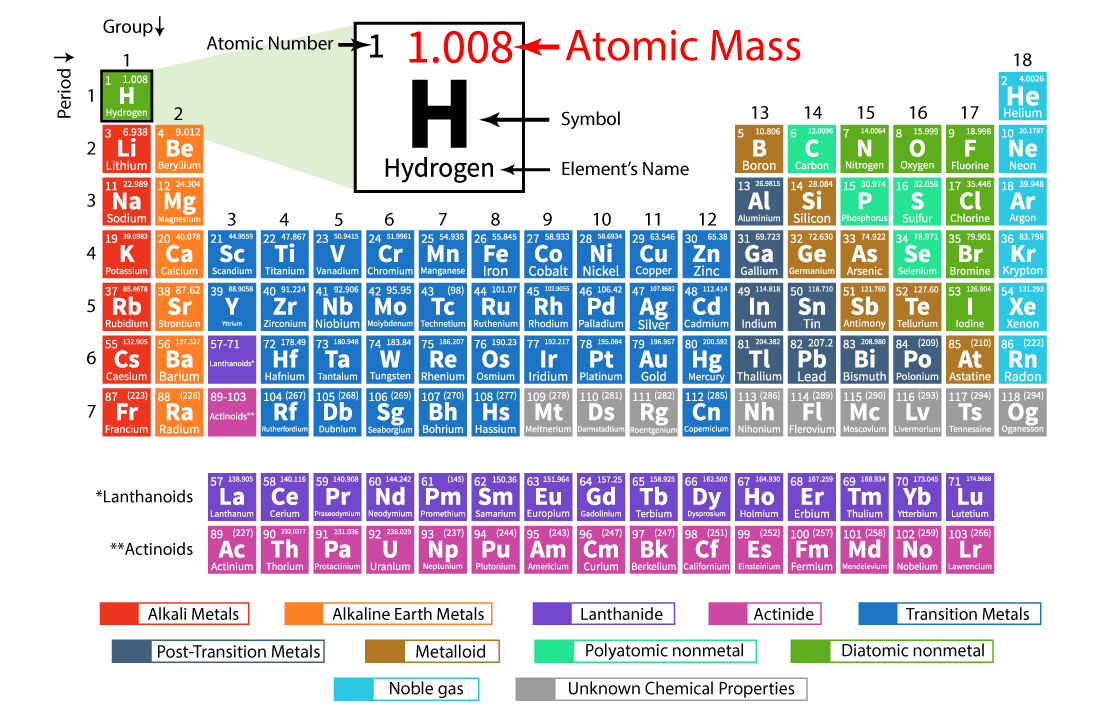
Phosphate rock (calcium phosphate) can be heated to 1200-1500 o C with sand (SiO 2) and coke (refined coal) to produced P 4 (vaporised), and after it condensed into a white powder under water (prevent oxidation in air):Ĥ Ca 5(PO 4) 3F + 18 SiO 2 + 30 C → 3 P 4 + 30 CO + 18 CaSiO 3 + 2 CaF 2 Phosphorus doesn’t react with Iodine (I 2): Reacts with Bromine with excess phosphorus: Phosphorus reacts with excess Bromine (Br 2), and forming phosphorus (V) bromide: PCl 3 (s) + Cl 2 (g) → PCl 5 (s) (phosphorus pentachloride) Phosphorus reacts with excess Chlorine (Cl 2), and forming phosphorus (V) chloride:Īnd reacts with Chlorine with excess phosphorus:Ģ P (s) + 3 Cl 2(g) → 2 PCl 3 (s) (phosphorus trichloride) Reacts with Fluorine with excess phosphorus, It forms: Phosphorus reacts with excess Fluorine (F 2), and forming phosphorus (V) fluoride: It doesn’t react with water and dilute non-oxidizing acids: Under controlled conditions (25% N 2, 75% O 2, 50 o C, 90 mm Hg), a mixture is formed, which containing phosphorous trioxide (actually tetraphosphorous hexaoxide, P 4O 6) Space Group Number: 2 Body Centered Cubic (BCC) Reactivity of PhosphorusĮlectron affinity: 71 kJ/mol Nuclear Properties of PhosphorusĪPhosphorus ignites spontanteously in air at room temperature, and forming phosphorus pentoxide (actually tetraphosphorous decaoxide, P 4O 10): Ionization energies: 1st: 1012 kJ.mol 2nd: 1907 kJ/mol 3rd: 2914 kJ/mol Sound Speed: 2130 m/s Atomic Properties of Phosphorus 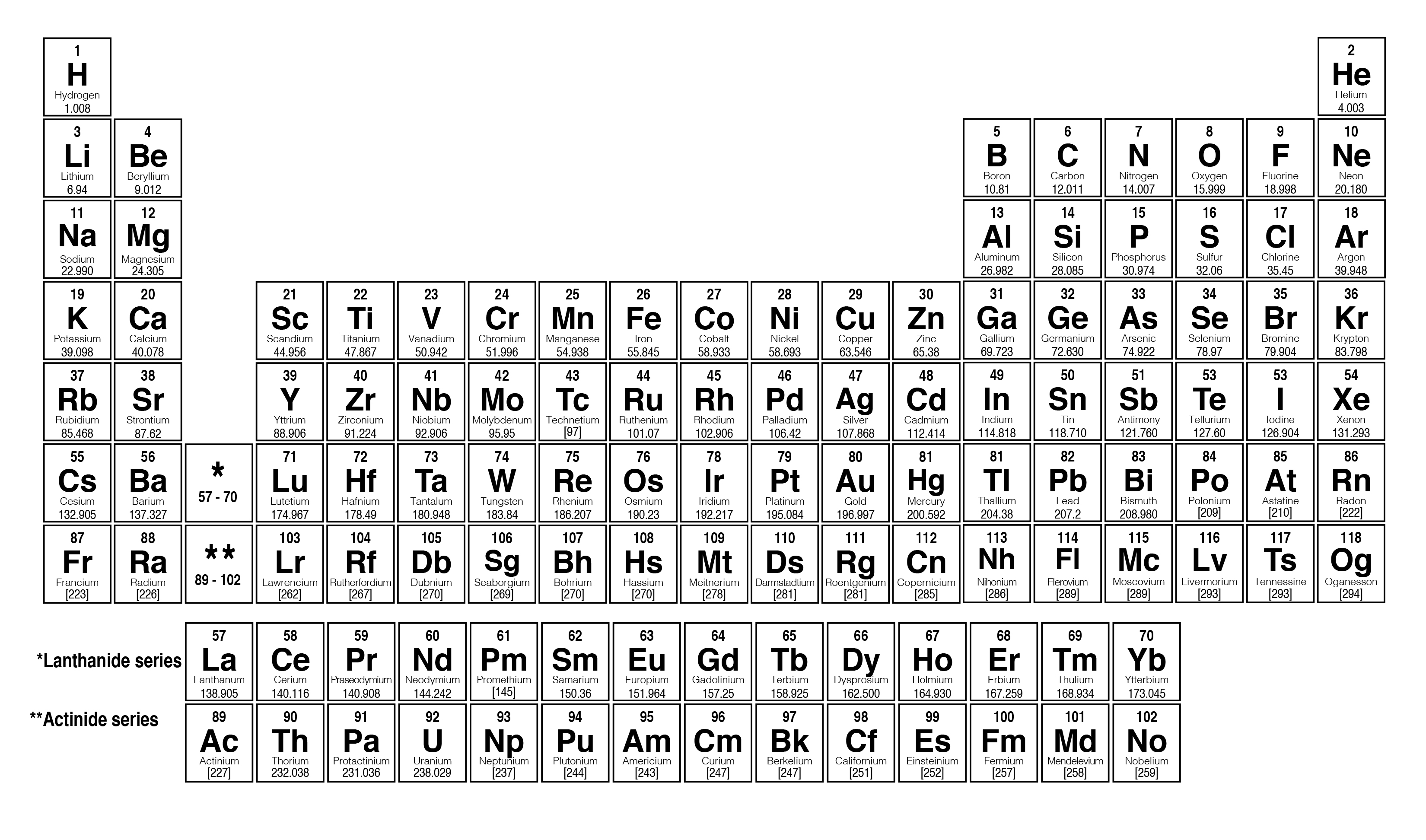
Molar magnetic susceptibility: -35×10 -9 m 3/mol Physical Properties of Phosphorusĭensity: white: 1.823 g/cm 3, violet: 2.36 g/cm 3, red: ~2.2-2.34 g/cm 3, black: 2.69 g/cm 3 Mass magnetic susceptibility: -0.113.x10 -9 m 3/kg Volume magnetic susceptibility: -0.0000206 Magnetic susceptibility (x mol): -20.8×10 -6 cm 3/mol Thermal conductivity: white: 0.236 W/(m∙K), black: 12.2 W/(m∙K) Electrical properties of PhosphorusĪ Electrical type: Conductor Magnetic Properties of PhosphorusĪ Magnetic type: Diamagnetic (white, violet, red, black)Ĭurie point: (above which Ferromagnetism vanishes) Molar heat capacity: white: 23.824 J/(mol.K) World’s Top 3 Reserve holders of PhosphorusĪllotropes: White phosphorus, Red phosphorus, Black phosphorusĪppearance: colourless, waxy white, scarlet, yellow, violet, red, blackĮlectron configuration: 1s 22s 22p 63s 23p 3 Phosphorus Electron Configuration Thermal Properties of Phosphorus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
